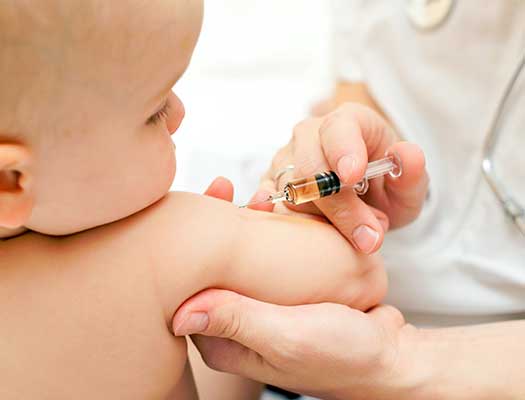
Chickenpox Outbreak in Williamsburg May Spread
The New York City Health Department is investigating an outbreak of varicella (chickenpox) in the Orthodox Jewish community of Williamsburg, Brooklyn. To date, there have been 75 reports of persons with varicella who became ill during or after March 2016. The median age of patients is 3 years (range 0 to 10 years); 72% were not vaccinated against varicella, and 14% had not yet received the recommended number of doses.
Clinical Presentation
Varicella is an illness characterized by a generalized, pruritic rash. It progresses from macules to papules to vesicular lesions and then crusts over. Lesions are usually most concentrated on the trunk. Persons who were previously vaccinated may develop varicella, but symptoms of infection are often mild; fewer lesions may develop, lesions may not be vesicular, and fever may be absent. It is important to maintain a high index of suspicion for varicella in patients presenting with a compatible illness, regardless of vaccination history. Infants, adolescents, adults, pregnant women, and immunocompromised persons are at risk for more severe disease and complications. Complications include pneumonia, bacterial infection of the skin and soft tissues, meningitis, encephalitis, birth defects, and death.
Transmission and Infection Control
Varicella is highly contagious. People with varicella are contagious beginning 2 days before rash onset until all lesions have crusted and no new lesions have appeared for a 24 hour period. People who are not immune to varicella are at risk for getting sick from 10 to 21 days after exposure, and they may be contagious as early as 8 days after exposure. In healthcare settings, providers should institute contact, droplet and airborne precautions. All healthcare workers should have evidence of immunity to varicella (see below).
Laboratory Testing
Because widespread vaccination has made varicella uncommon, diagnosis based on history and physical examination may be difficult, and laboratory confirmation is important. The preferred diagnostic test is polymerase chain reaction (PCR) of skin lesions (vesicles, scabs, maculopapular lesions). PCR testing can be performed at commercial laboratories. For additional information on specimen collection, see: www.cdc.gov/chickenpox/hcp/lab-tests.html. IgM serologic testing is less sensitive than PCR.
Vaccination
Please ensure that your patients and staff are up to date with varicella vaccine. Two doses of the vaccine are up to 98% effective at preventing chickenpox. Varicella vaccine should be given to children at 12 months of age with a second dose at 4 years of age. Two doses of vaccine are recommended for older children and adults who do not have evidence of varicella immunity. Adults who do not know if they had varicella and who do not have their immunization records should either be vaccinated or can have varicella IgG titers checked to confirm whether they are immune. Vaccination histories of children can be obtained through the Citywide Immunization Registry atwww.nyc.gov/health/cir or by calling 347-396-2400.
Evidence of immunity
People exposed to varicella should be assessed for presumptive evidence of immunity:
1) Documentation of age-appropriate vaccination with varicella vaccine,
2) Laboratory evidence of immunity or laboratory confirmation of disease,
3) Birth in the United States before 1980 (except for health-care personnel, pregnant women, and immunocompromised persons), or
4) Health-care provider diagnosis or verification of a history of varicella or herpes zoster.
People exposed to varicella who do not have evidence of immunity should remain home while at risk from getting sick, through 21 days after their last exposure.
Post-exposure Prophylaxis
Varicella vaccine should be administered as post-exposure prophylaxis to persons without immunity to varicella within 3 days of an initial exposure to prevent infection. Vaccination beyond this time frame, while still important for protecting a person from future exposures, may not protect against exposures that have already occurred. These people would still need to be excluded from their school or workplace. Varicella zoster immune globulin (VariZIG) post-exposure prophylaxis is reserved for persons at high risk for severe disease who lack evidence of immunity to varicella and for whom varicella vaccine is contraindicated. This includes immunocompromised or pregnant persons without evidence of immunity, newborn infants whose mothers have symptoms of varicella around the time of delivery, and certain hospitalized premature infants. People who receive VariZIG should not receive measles- or varicella-containing vaccine for at least 5 months after receiving VariZIG. Information about ordering VariZIG, indications, dosing is at: www.cdc.gov/mmwr/preview/mmwrhtml/mm6228a4.htm.
Treatment
Antiviral treatment is not recommended for routine use in otherwise healthy children with varicella. Oral acyclovir or valacyclovir should be considered for persons at increased risk of moderate to severe varicella, including unvaccinated persons aged >12 years. Intravenous acyclovir is recommended for pregnant women with serious varicella complications and immunocompromised persons. For additional information on treatment, please refer to the Red Book, at http://redbook.solutions.aap.org/.











Anonymous
They should have a refua shelaima
But this is not a reason to get vaccines
Milhouse
It certainly is. An unimmunized child’s risk of dying from chicken pox is far higher than that of its being harmed in any way by the vaccine.
buchurel
At least all those kids are safe from the horrible diseases associated with vaccines, like autism and scrofula!
Anonymous
Vaccinations save lives. I hope the above comments are jokes
pregnant woman, cancer patients, very young kids
pregnant woman who never had chickenpox is very likely to abort if she gets chickenpox.
cancer patients, very young kids and old adults can die from it.
if you’re meshuga enough not to vax your kid now, once he gets chickenpox that might develop in later years into shingles or herpes. if you dont vax by listening what hippies have to say instead of doctors, that’s the great gift you’re giving your child: a painful, horrible and disgusting herpes.
1990's
We all got chickenpox as kids. This is fear mongering. You do not need to vaccinate for chickepox. Many people have chicken pox parties to get their children exposed which causes immunity for life. The only time it is dangerous is when a child starts hitting puberty and never got chickenpox. Only then should they get the vaccine .
Milhouse
Chickenpox is not as serious as a lot of other diseases, but it can still be serious and even fatal r”l. Yes, we all got it and survived — that’s why we’re here to talk about it, but what about the kids who didn’t survive? They were just like us, but they’re not here to tell their stories. However small this risk — and it is very small — it is still orders of magnitude greater than the risk from the vaccine.
But even the vast majority of children who get through chickenpox without any serious consequences still suffered for days. Now that there is an alternative, what kind of sick sadistic parent would deliberately subject their child to the discomfort of chickenpox when there is an alternative? That they are also subjecting their child to a small but real risk of serious harm just makes it worse.
Chickenpox parties were a good idea before the vaccine was developed. Those who still have them today are irresponsible and ignorant, and will have to account to Hashem for their negligence with the precious children that He gave them.
Experienced mom
Educate yourself and do research before posting comments. My son developed complication from a light case of chicken pox, before the shots came out. He was hospitalized and out of commission for 6 weeks. The virus entered his nervous system. I wish the shots were available then. Children or unborn exposed to measles were born deaf or had some other birth defect. Vaccination saved many children.
By now most people are aware that the person who started the rumor that vaccinations cause autism, had no real studies to back up his claim. If you want to be cautious ask the dr. to separate the shots.
stop being a fool
its funny how people can claim vaccines cause autism based off of one bad study that was published and later taken back by all but one of the people that did it.
it seems that people are willing to trust science when it tells them vaccines are dangerous, but unwilling to listen when the entire scientific community tells them its not. if you believe that vaccines cause autism then you need to read up about the study published in the lancet and on how scientific studies work and are accepted.
To #4
not jokes at all. How well do you know the ill effects of vaccines? Chicken pox – up until a few years ago – was something most kids got. Uncomfortable to deal with, but we all got it and B”H survived! Many who get the chicken pox vaccine end up getting shingles later on in life – a much worse choice.
Milhouse
There are no significant ill effects of vaccines.
And yes, we all BH survived chickenpox; what about the ones who LA didn’t? They’re not here to give their opinion.
to number 1
people like you should be incarcerated because putting children at risk like this is a SIN. Do you read or have any kind of education?Ignorance is not bliss.
oy vey
what chemicals are in the vaccine?
lets see who wants that floating in you body for years?
Milhouse
There’s no reason not to want chemicals “floating in your body”. Your body is made of chemicals. So is everything you eat, touch, or interact with in any way. There’s no reason to suppose the ones in vaccines are any more harmful than the ones in the healthiest foods.
Chemicals
Newsflash: your entire body is FULL of chemicals — all produced by… your body! Your body itself is largely composed of CARBON, even NITROGEN (which is explosive!)
We are not the Amish, who shun electricity and cars, nor the Adventists who refuse blood transfusions. Since when did “natural” become a higher value then the chiyuv deOraisa of ushmartem es nafshoseichem?
#13
Are you abt 10 yrs old and/or did you finish elementary school? let me guess what school you may have gone to- did they teach any science courses?Do you know how to read a study and see the conclusions?Can you even read?If the answers are negative, why are you even making comments about a very serious subject??????
Don't put your child at risk.
My son had chicken pox as an infant years before the vaccine was recommended. He spent four weeks in the hospital in critical condition. Yes most children get over it with little side effects, like my other children did but unfortunately even one fatality is too much risk.
Anonymous
A classmate of mine died at age 11 from chickenpox. She had complications resulting in pneumonia which a risk of that age group.
Educated
Maybe if most Lubavitchers got more than a 6th grade education, there would be fewer incredibly ignorant people making stupid irresponsible comments like 1, 3, 6, 10.. There might be more vaccinated children and adults, and as a result, fewer cases of varicella, rubella, mumps, meningitis, etc. More of us would be alive, healthier.
But, nebuch, people would rather believe nonsense fairy tales from some internet celebrity about autism and “chemicals in the body” and other narrischkeit, and refuse to vaccinate.
It is high time to require proof of vaccination to allow kids in cheder, in camp, etc.